Do You Own an Epinephrine Auto-Injector? Read This!
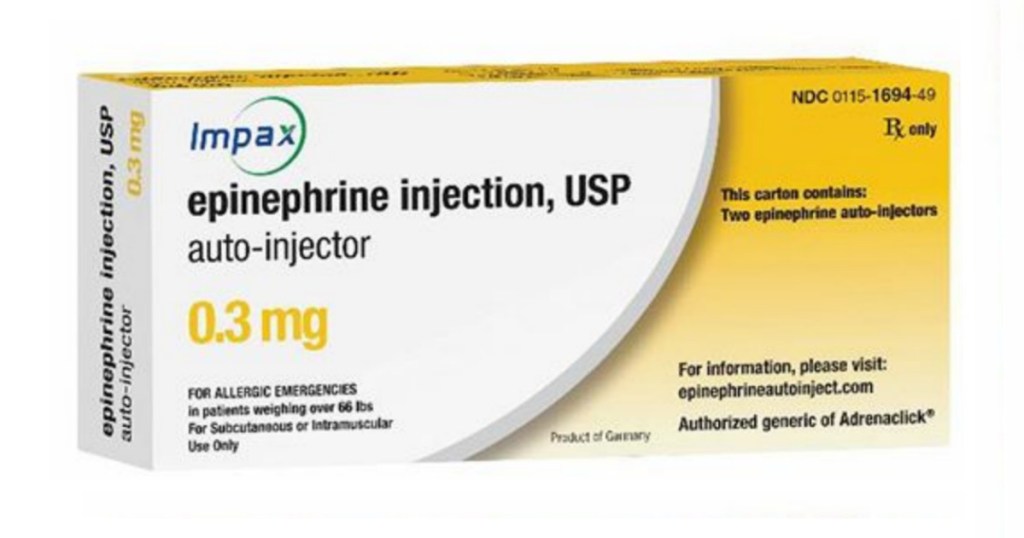 The Food and Drug Administration is suggesting that anyone who received an Amneal Pharmaceuticals LLC or Impax Laboratories epinephrine injection, USP auto-injector 0.3 mg device after December 20th, 2018 to inspect the device to make sure it has a yellow stop collar.
The Food and Drug Administration is suggesting that anyone who received an Amneal Pharmaceuticals LLC or Impax Laboratories epinephrine injection, USP auto-injector 0.3 mg device after December 20th, 2018 to inspect the device to make sure it has a yellow stop collar.
This stop collar controls the dose of medicine administered so if you do not see the yellow stop collar, there is a risk of overdose.
The possible faulty devices have NDC numbers 0115-1694-30 on the case and 0115-1694-49 on the carton. The lot numbers and expiration dates are listed in a letter to health care providers on the FDA website.
Follow the steps here to confirm that the yellow stop collar is present.
If the yellow stop collar is missing, call the Amneal Drug Safety Department at 1-877-835-5472 to return the defective devices for a replacement. You should also contact your pharmacy about a replacement epinephrine auto-injector before returning the defective device.
Do you have a life-threatening allergy? Get this EpiPen Alternative for FREE!






Wow as a school nurse it makes me wonder how long these have been out………scary. I wonder where they were made………….we need to bring drug and medical suppliers back to the USA……….it’s well worth the cost increases………these were made in Germany. It’s on the front of the box.
it says they have been out since Dec. of 2018
Thank you so much for this post. I just got off the phone with CVS and they are replacing ASAP. I appreciate this post!
Oh good! You’re welcome, Emily! SO happy this was helpful. I have to call CVS today about mine too!
Thank you so much for posting this. As a mom with a child with severe food allergies, I really appreciate you raising awareness about these faulty pens.
You’re SO very welcome, Becca! 🤗
Thanks for posting this!! I had to go check my pens and they DO have the stop collar. This also reminded me to refill my prescription as well.
You’re welcome!
Another allergy mom posting a big THANK YOU for this post!
You are SUPER welcome!
Thank you so much for posting this! I’m gonna have to recheck my sons epi pens
You’re welcome, ammber!
Well this is a little terrifying. I shudder to think how many kids had to overdose before they realized their mistake. I used to work in quality control at a pharma company and this is the stuff you have nightmares about.