Abbott Recalls FreeStyle Glucose Monitoring Systems Due to Fire Risk
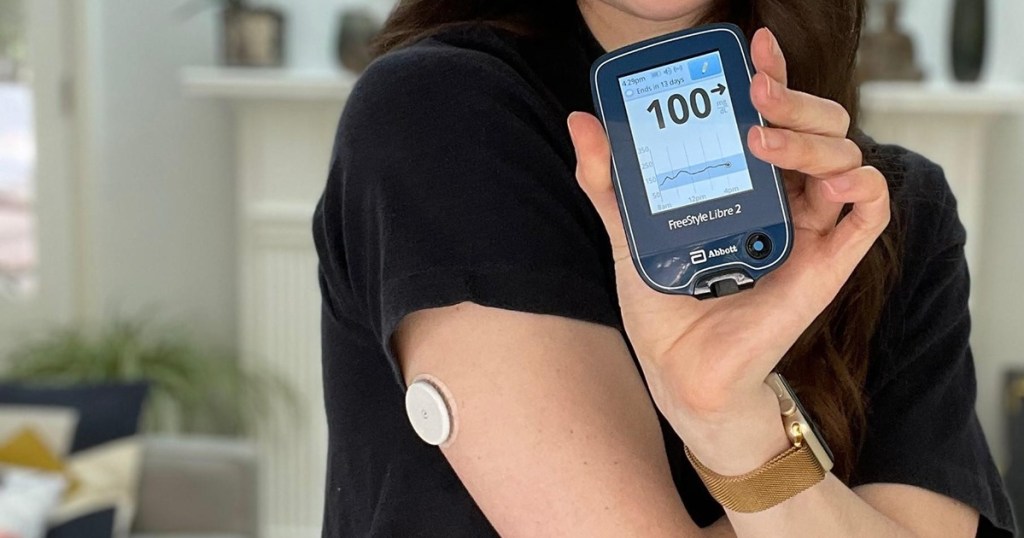
Abbott has issued a recall of the FreeStyle Libre, Libre 14-day, and Libre 2 Flash Glucose Management Systems’ reader devices. These doctor-prescribed devices help people manage their diabetes by tracking glucose levels so treatment can be adjusted as needed.
The recalled FreeStyle Libre devices use rechargeable lithium-ion batteries, which may get extremely hot, spark, or catch fire if not properly stored, charged, or used with the Abbott-provided USB cable and power adapter. This risk is also increased by other misuses, including exposure to liquids, damage, and introduction of foreign material into the ports.
If the battery swells or overheats, users may be exposed to extreme heat or fire, which can cause serious injuries or death. Additionally, users may miss a critical diabetes treatment if the system cannot be used after it’s damaged by heat or fire. There have been 88 incidents (including at least seven reports of fires and one injury) related to this issue.

No FreeStyle Libre Readers are being physically recalled at this time.
If you have the Abbott-provided USB cable and power adapter AND you are not experiencing any of the problems from the FDA’s list, you can continue to safely use your device. The Abbott-provided USB cable and power adapter limits the power provided to safely charge the battery.
If the Reader is damaged or a replacement USB cable or power adapter is needed, please call Abbott’s Customer Service at 1-855-632-8658 to request a replacement. The official recall notice contains detailed information regarding how to check your device for damage.
Note that this medical device correction does not affect the FreeStyle Libre family of sensors.




Comments 0